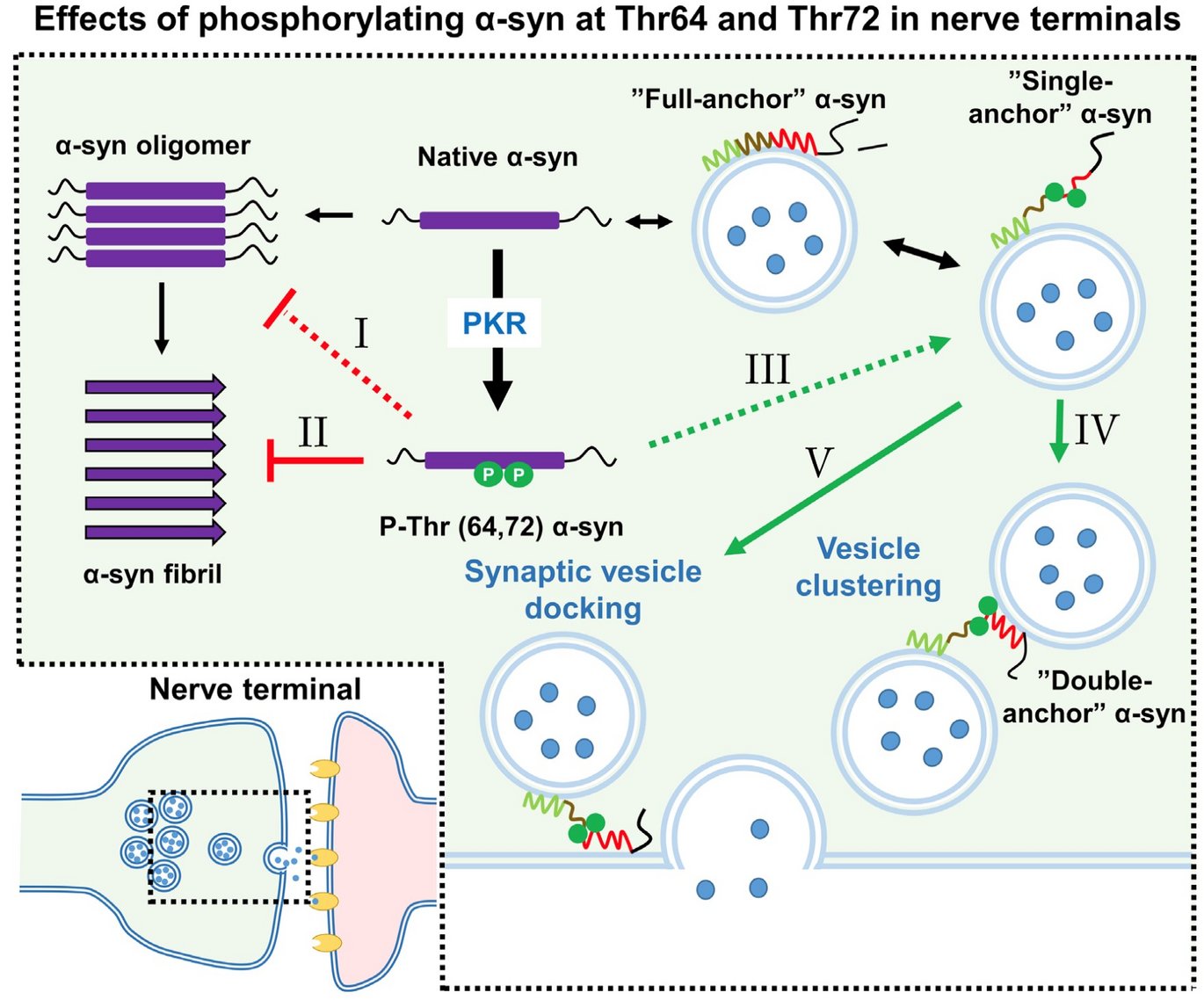
Effects of phosphorylating α-syn at Thr64 and Thr72 in nerve terminals
Collectively, we demonstrate for the first time that the binding between alpha-synuclein and membranes can be regulated by phosphorylation by the kinase PKR, identifies the responsible phosphorylation sites, and demonstrates it can modulate the binding between two vesicles by favoring the “double anchor” vesicle-binding mode. Moreover, phosphorylation inhibits the pathological aggregation of alpha-synuclein in cells and brain tissue.
This collaborative study was conducted between friends and experts from the USA, China, Switzerland, United Kingdom, Italy, and Sweden situated at the University of Washington School of Medicine; School of Life Sciences Brain Mind Institute, Lausanne; University of Cambridge; University of Naples, Lund University, Zhejiang University School of Medicine and the First Affiliated Hospital.
Published in PNAS Nexus, Volume 1, Issue 5, November 2022, pgac259.
Read the full article here
