A new powerful toolbox may solve big questions in small synapses
The development of new cutting-edge techniques allows researchers to delve into the molecular architecture of one of the smallest structures in the brain – the synapses. This innovative approach is expected to yield important insights into the onset of several major brain disorders.
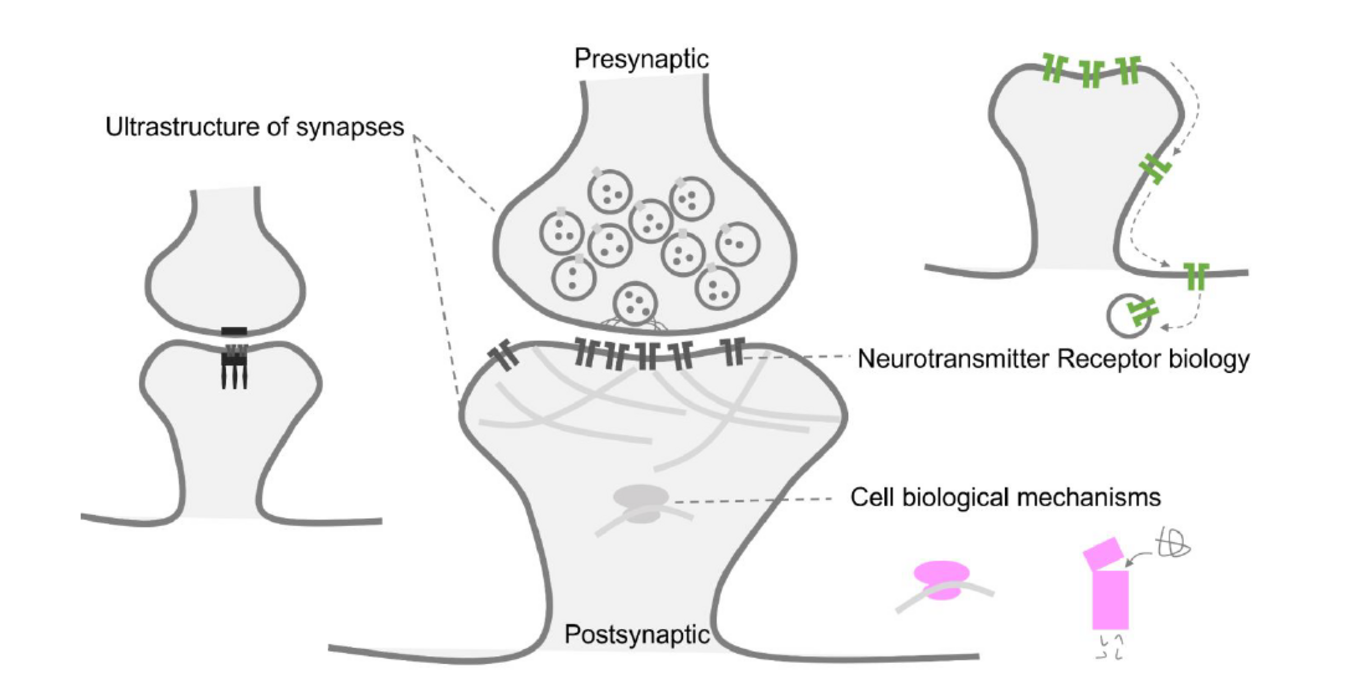

Synapses between neurons are responsible for processing and storing information in the brain. Unlike many other organs in the body, synapses can change in shape and strength throughout life. As such they play a huge role in the homeostasis of the brain and proper brain function.
For this reason, pathology at synapses can carry fatal consequences and it is well-known to be a common cause of major brain disorders such as Parkinson’s Disease and Schizophrenia.
Up until now, detailed investigation of the physiology of synapses using conventional light microscopy has been nearly impossible because of their sizes. With a measure of only a few hundred nanometers in diameter, they are comparable to the wavelength of light.
“Light cannot resolve objects that are below its wavelength. This is called the diffraction limit. Due to this diffraction limit, conventional microscopy cannot see clearly what’s inside synapses,” explains Associate Professor and DANDRITE Group Leader, Chao Sun.
In a new perspective just published in “The Journal of Physical Chemistry”, he describes a set of cutting-edge technologies that are so powerful they can visualize individual molecules at synapses.
“There is an increasing repertoire of techniques that allow us to resolve individual molecules in situ at synapses, such as super-resolution microscopy, cryogenic electron microscopy and tomography, and single-particle tracking. These techniques make it possible for us to analyze the distribution, quantity, dynamics, structure, and function of individual molecules that reside inside synapses,” Sun states.
These in situ single-molecule techniques are beginning to transform our averaged knowledge of a synapse, promising exciting discoveries in subcellular biophysics of synapses and single-synapse proteomics. In essence, these techniques serve as a crucial gateway to unlocking the inner workings of synapses, enabling a deeper understanding of their biological mechanisms in both health and disease contexts.
“With these techniques, many exciting new questions and challenges have emerged. This will undoubtedly lead to new findings in the coming years,” Chao Sun concludes.
